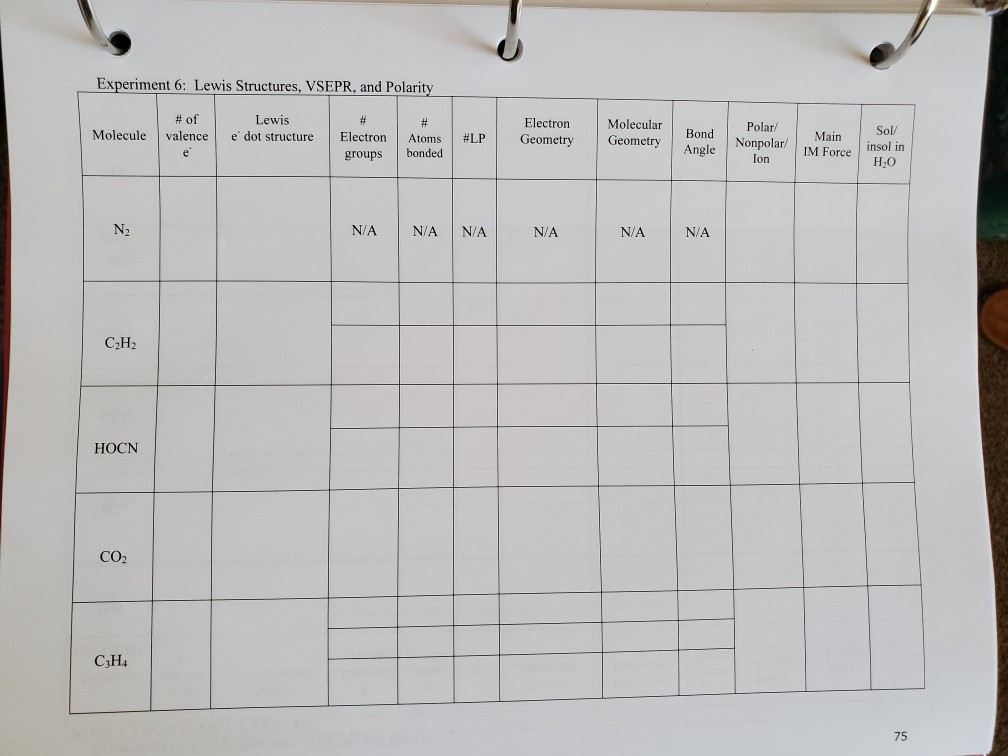
For each of the following molecules, (1) draw the correct Lewis structure; (2) label each polar covalent bond with a dipole moment arrow; and (3) determine if the molecule is polar or
Solved: Chapter 3 Problem 78CP Solution | Masteringchemistry -- Standalone Access Card -- For General, Organic, And Biological Chemistry 1st Edition | Chegg.com

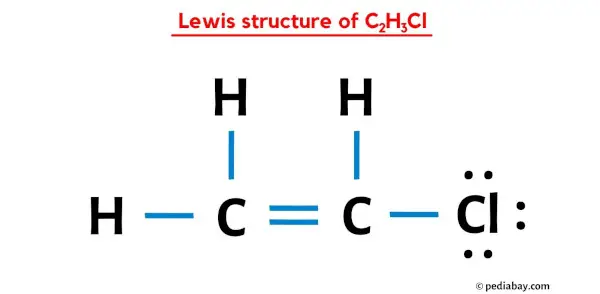
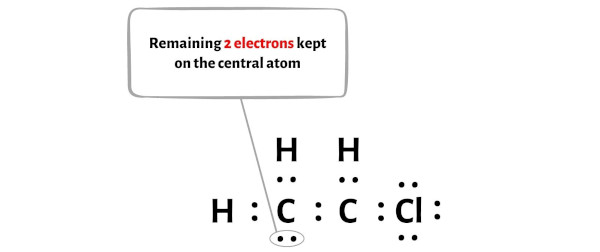
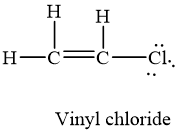
SOLVED: Determine whether each molecule is polar or nonpolar. Question 13 options: C2H3Cl HONO HCOOH C2H4 O2 HOCN C2H2 N2 1. polar 2. nonpolar

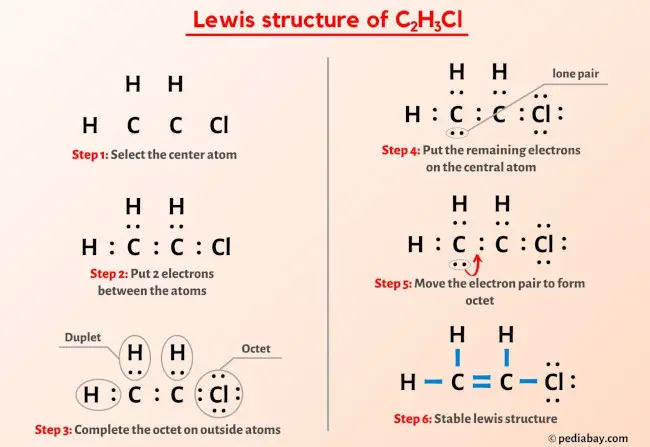
Draw a Lewis structure for C2H3Cl Show all unshared electron pairs None of the atoms bears a forma - YouTube
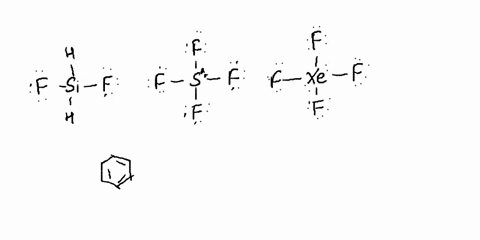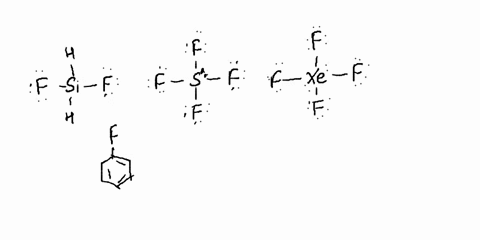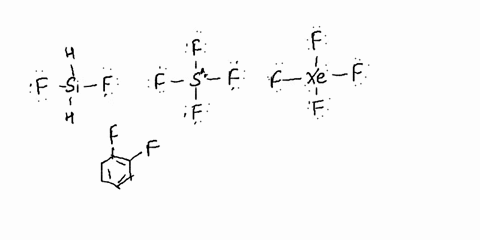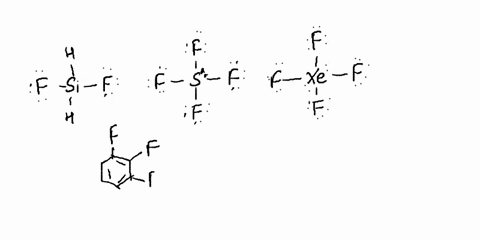
SOLVED: Determine whether each molecule is polar or nonpolar. Question 13 options: C2H3Cl HONO HCOOH C2H4 O2 HOCN C2H2 N2 1. polar 2. nonpolar
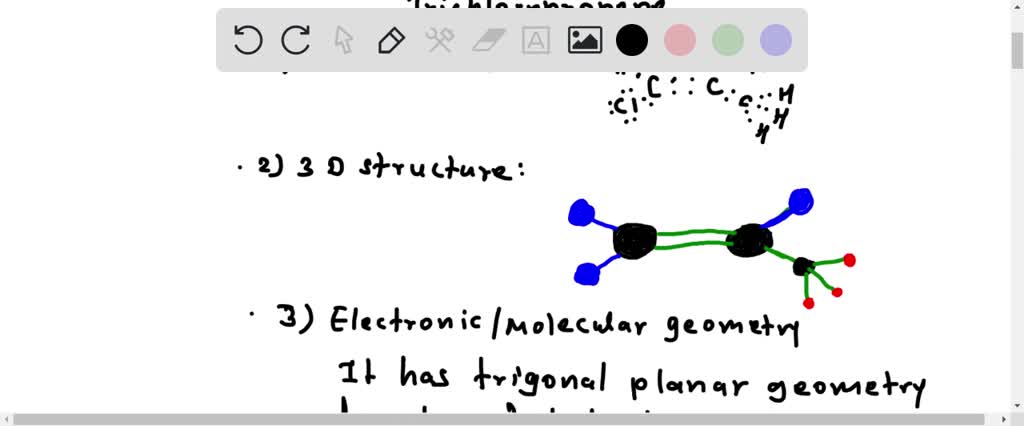
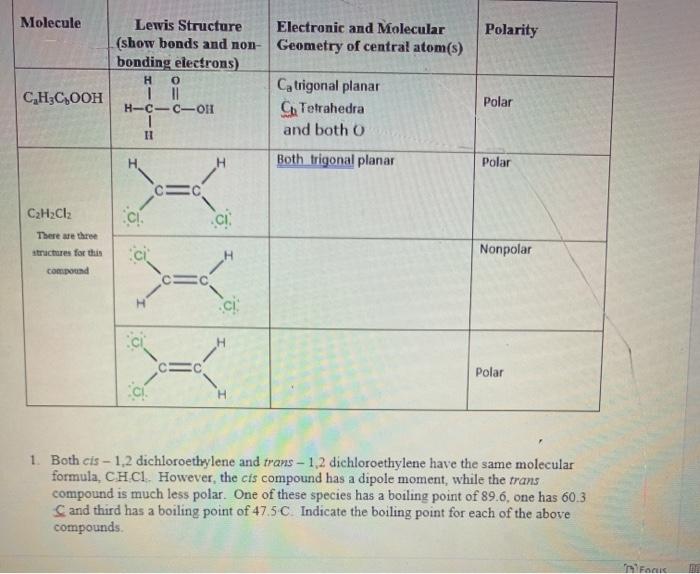
SOLVED: Draw and write C2H3Cl3 1. Lewis Structure 2. 3D structure 3.Electronic Geometry / Molecular Geometry 4.Hybridization 5. Polar or Non-polar

Biacetyl and acetoin are added to margarine to make it taste more like butter. Complete the Lewis structures, predict values for all C-C-O bond angles, and give the hybridization of all the

SOLVED: Determine whether each molecule is polar or nonpolar. Question 13 options: C2H3Cl HONO HCOOH C2H4 O2 HOCN C2H2 N2 1. polar 2. nonpolar
A student places a mixture of plastic beads consisting of polypropylene (PP) and polyvinyl chloride (PVC) in a 1.0 L beaker cont